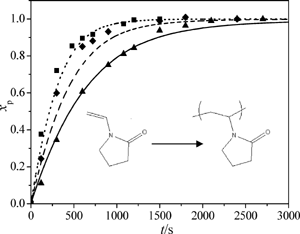
Aqueous-phase free-radical batch and semibatch polymerizations of NVP have been carried out with varying initial monomer and initiator concentrations. The rate of conversion was observed
to increase as the initial monomer concentration was lowered, a result explained by the dependence of the propagation rate coefficient, kp, on monomer concentration. A kinetic
model with termination and conversion-dependent kp rate coefficients taken from independent studies provides a good description of the conversion profiles. A reasonable
representation of polymer molecular-weight averages and the complete MWD was obtained by assuming that the rate coefficient for transfer to monomer also varies as a function of monomer
concentration and that a small amount of chain transfer to polymer occurs.
